While we are very familiar with turbine and hydraulic oils undergoing oxidation, the question has arisen, “Can transformer oils also experience oxidation?” Yes, like most oils, transformer oils can experience oxidation however, there are certain factors which affect their rate of oxidation such as its load factor.
The oxidation process occurs when a chemical reaction occurs between the organic hydrocarbon chain and oxygen. This reaction can result in the formation of reactive oxidation compounds which will begin attacking the oil’s base stock and can form polymers. Similar to regular oils, these oxidation products or contaminants can cause increased heating and corrosion. They can also deposit on windings, tank walls and cooling ducts which hamper free circulation.
Oxidative accelerators
Oxidation can be accelerated in transformer oils. Here are three parameters which can act as oxidative accelerators:
*Temperature stress: local hotspots or an overall high operating temperature can impact the rate of oxidation (as per Arrhenius rule – for every 10°C rise, the rate of oxidation will double and the oxidative oil life will be halved). This is significant for transformer oils.
*Solid contamination: accelerates oxidation as a catalyst. This is especially regarding the reaction products and fibers from the insulating paper inside of these electrical pieces of equipment.
*Water or wear: moisture or water contamination can partially be absorbed while oil is being oxidized. These can be absorbed into the oil degradation products and negatively influence the surface tension of the oil. The heavy molecules of water and degradation products can move to the paper insulation and reduce the insulation resistance.
These oxidative accelerators will increase the fluid degradation especially if they work in combination. For every 10 degree rise in temperature, the life of the paper insulation is halved (due to aging). Similarly, when the moisture content doubles, the paper life is reduced by half due to the loss of mechanical strength. Let’s investigate the effects of water on transformer oils.
The dangers of water in transformer oils
Transformer oils have a dual functionality namely to lubricate and insulate the equipment. Water acts as a contaminant in transformer oils and can easily enter transformer systems due to the presence of moisture. In microscopic or larger volumes, water can harm the insulations which are immersed in the oil. Water is an unwanted substance and is directly responsible for many electrical breakdowns. As such, the presence of water in this type of oil can have serious effects.
There are a couple of ways in which water can get into transformer oil systems. These include:
- leaking gaskets
- incorrect lubricant handling techniques
- reaction between insulating paper and oil degradation products.
In these cases, the water can be under free form, as a separate layer or in the dissolved phase.
In addition to affecting the electrical equipment, the presence of water in transformer oils can also lead to:
- reduction of the strength of the oil
- loss of insulating characteristics
- dielectric breakdowns (at sudden high loads, the water can boil off the conductor surfaces and the vapour bubbles can cause dielectric failures)
- increase in oxidation rate and creation of oxidation products (in combination with water, these will prefer the cooler parts of the transformer oil and can increase corrosion).
- decrease in insulation resistance (oxidation products can find their way to the paper insulation causing it to expand and alter the mechanical pressure of the transformer clamping system).
Overall, water can have detrimental effects on transformer oil systems and these oils should be kept clean and dry to increase the longevity of the oil.
Understanding Antioxidants and their roles
Antioxidants are the protectors of base oils. They can perform this role by either scavenging (alkyl and peroxy) radicals or decomposing hydroperoxides into stable products. There are two main categories of antioxidants, namely;
*Primary antioxidants: these remove radicals and are better known as radical scavengers. The characteristic types of primary antioxidants are amines and phenols. In turbine oils, these primary antioxidants are typically in concentrations of 0.3 -0.7 wt%.
*Secondary antioxidants: these eliminate peroxides (hydroperoxides decomposers) and form non-reactive products which do not participate in further oxidation of the lubricant.
*Mixed antioxidants systems: in these systems, two or more antioxidants are added to a fluid. Antioxidants are often used in synergistic mixtures to achieve an extended lubricant life. In this mixture, one of the antioxidants sacrifices itself in preservation and regeneration of the other. One example is the use of amines and phenols whereby the phenols deplete early in during oxidation whilst the amine concentration remains constant during this time. Another example can be the combination of a primary and secondary antioxidant where both radicals and hydroperoxides will be removed.
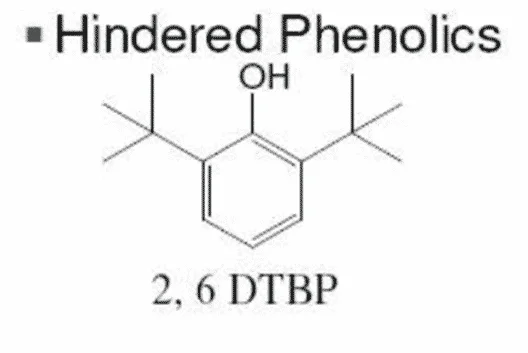
There are two main types of transformer oils as shown below:
- Non-inhibited transformer oils (these have no added antioxidants)
- Inhibited transformer oils or fluids which have a specific concentration of antioxidants such as DBP or DBPC which are phenolic types of antioxidants.
Monitoring the Remaining Useful Life
During oxidation, organic or weak acids are produced. These can result in corrosion damage in the long term. However, these acids may not be in a very high concentration to be flagged by the Acid number test. On the other hand, heavy deposits can also be produced during the polymerisation stage of oxidation. These can lead to an increase in viscosity but a viscosity test may not be able to detect this increase until after the polymerization has occurred and deposits have been circulated through the system.
The new generation of lubricants do not conform to a linear form of degradation (as it used to in the past). Instead, once the antioxidants have been depleted, Group II & III base oils have a less natural oxidative stability than Group I oils and they degrade very quickly. As such, there is usually no warning to indicate the onset of degradation and generation of deposits. While these new generations of lubricants can last longer and perform better they can fail rapidly as shown below.
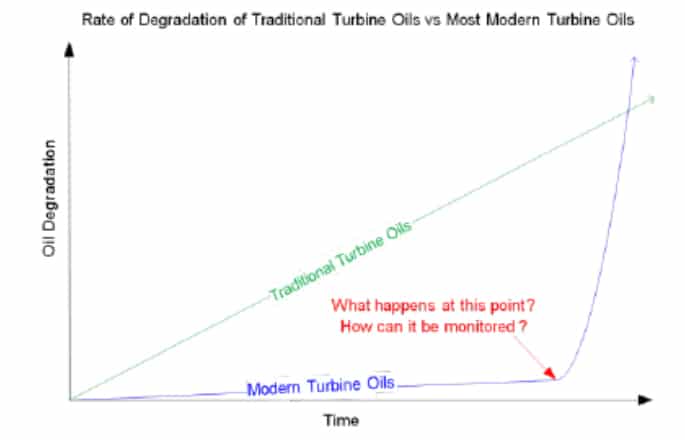
Therefore, a test to actively monitor the antioxidant concentration can assist in warning operators about the onset of oxidation. The RULER (Remaining Useful Life Evaluation Routine) test is one such test which can actively monitor these concentrations. This test utilizes linear sweep voltammetry to detect these concentrations which is an ASTM approved technique.
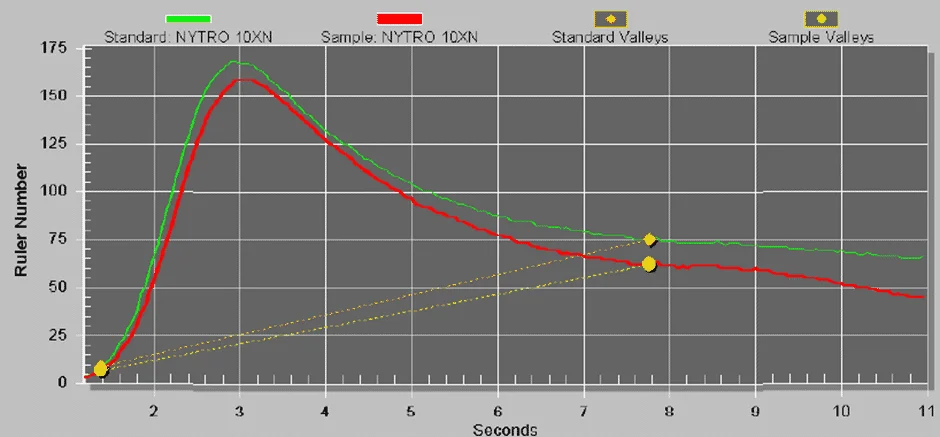
The method for ASTM D6810 specifically targets the measurement of phenolic inhibitors in oils. Additionally, ASTM has also approved a second method, ASTM D6971 which offers the ability to monitor the concentration of phenolic and aromatic amine antioxidants in non-zinc-containing turbine oils. The following is a voltammograph from a mineral type of transformer oil containing a phenolic type of antioxidant.
Case Study for Transformer oil
Transformer oils can consist of different mixtures of paraffinic, naphthenic and aromatic compounds. New generation (or hydrotreated) transformer oils undergo a higher degree of refining which results in a reduction of sulphur and polyaromatic content. These aromatics are important for non-inhibited transformer oils since they act as natural antioxidants. On the other hand, inhibited transformer oils can improve their oxidation stability by adding a suitable inhibitor (such as phenols).
The figure below shows a typical graph from a mineral transformer oil which shows two extremes of condition based on the presence of the remaining phenolic antioxidants using the RULER test.
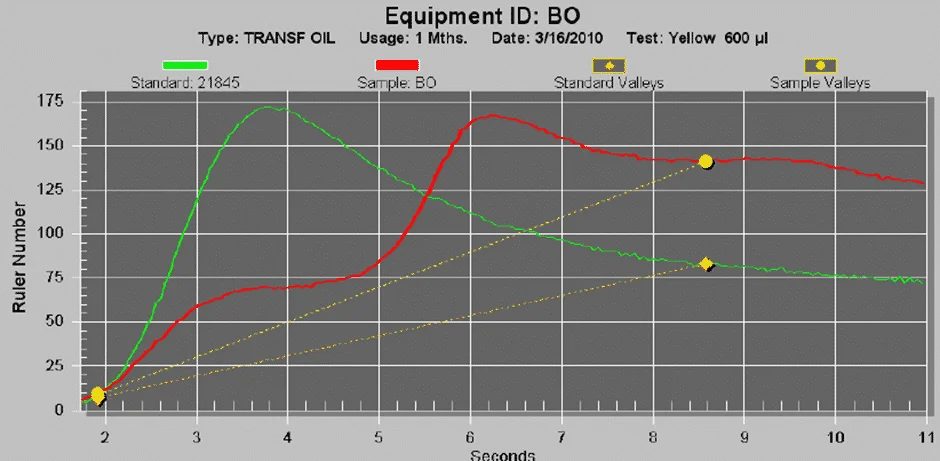
The analytical test data such as the Acid Number and Gas Chromatography analysis are in line with the results shown in the RULER test.
Overall, a transformer oil can become oxidized, just like a regular oil. However, the effects of oxidation on the system may differ slightly compared to oils used in rotating equipment or hydraulics since the oil has a dual role of insulation and lubrication for these electrical pieces of equipment. The disastrous effects of water for these types of oils were also noted; transformer oils should be kept clean and dry to improve their longevity. When determining their expected useful life, the RULER test is very helpful to detect the presence (or absence) of antioxidants.































